
Problem 14-127 |
For Al(NO3)3, NO3- is a spectator ion (conjugate base of a strong acid HNO3).
Therefore, in solution this compound will generate Al3+.
Small ion with high charge density in water is highly unstable:
Al3+ + 6H2O 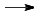 [Al(H2O)6]3+
[Al(H2O)6]3+
[Al(H2O)6]3+  [Al(H2O)5OH]2+ + H+ with Ka = 1.4 × 10-5
[Al(H2O)5OH]2+ + H+ with Ka = 1.4 × 10-5
This ion behaves like an acid in water (releasing one proton)