Problem 4-12 |
This is a stupid question from the book... Sorry!
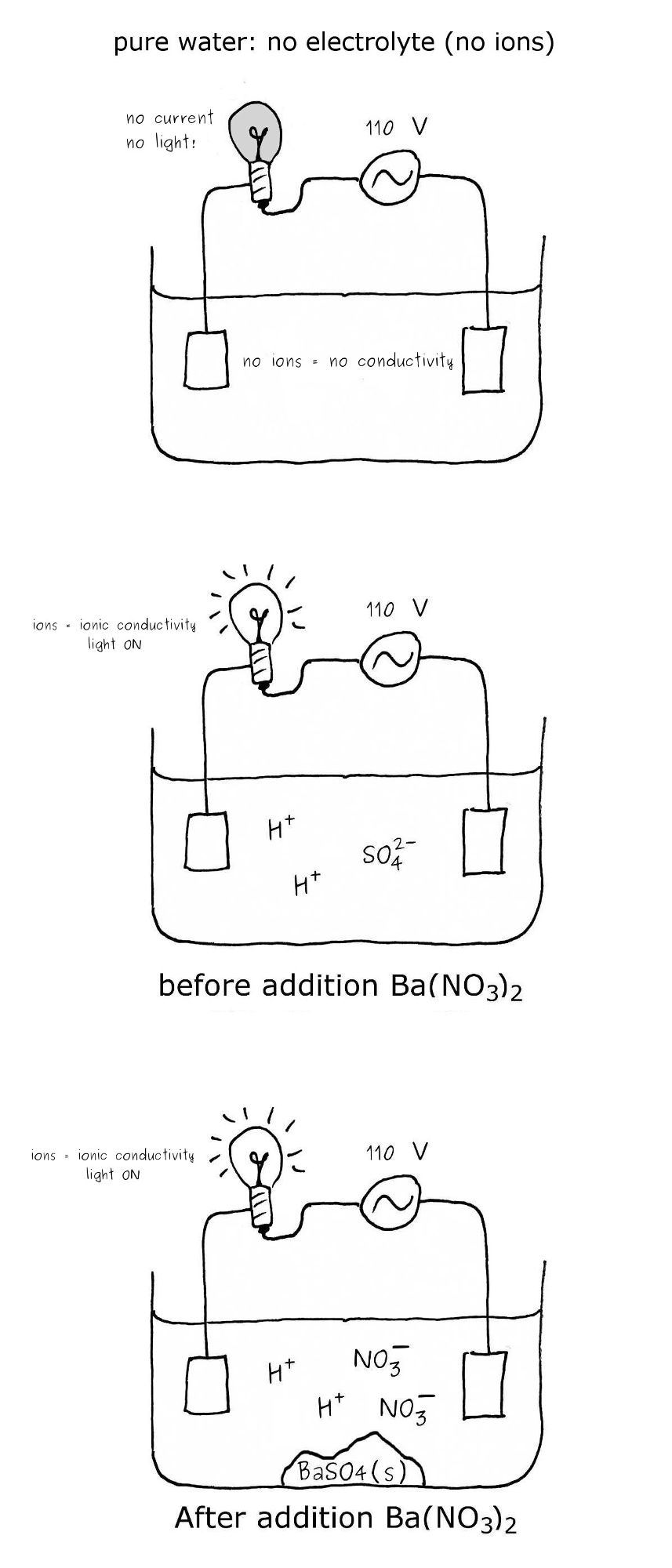
The bulb glows due to the presence of ions in the solution. Ions are “charge carriers” completing the electrical circuit.
If Ba(NO3)2 is added, it will precipitate the SO42- ions. However, each sulfate ion will be replaced by two nitrate ions (2 charge carriers).
The lightbulb will not dim. In fact, it is the opposite.
None of the dilute salt solution proposed in this question will decrease the intensity of the lightbulb...